Communication
International Collaborations and Missions
The latest international news, collaborations, events, and missions in life sciences and health.
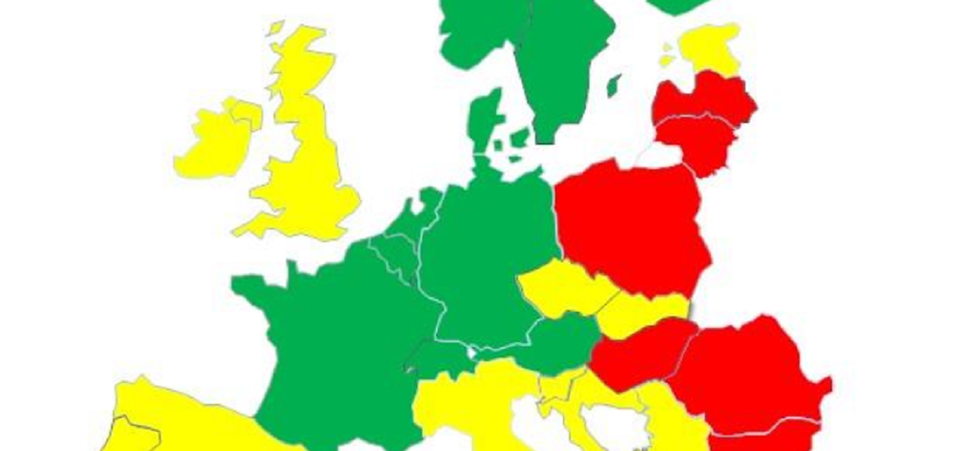
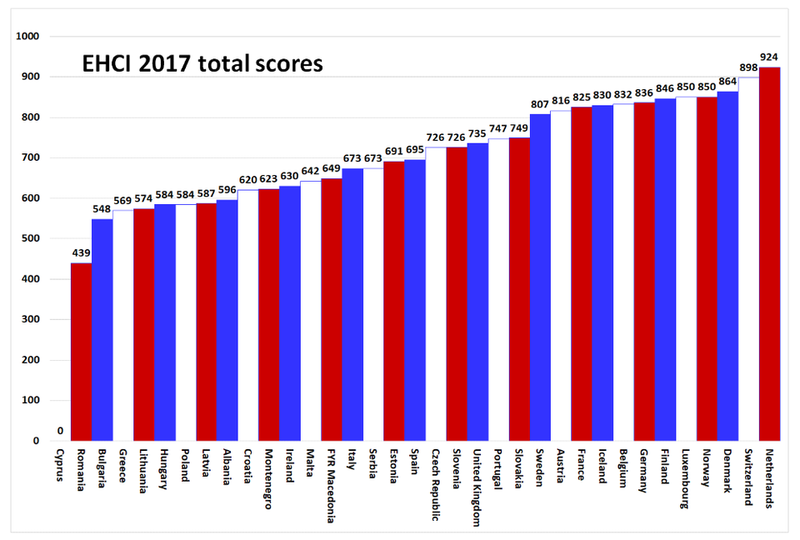
The position of the Netherlands according to the European Health Consumer Index
Read more ›

Join us for the BIO International Convention 2018
Read more ›

Join the second edition of the Global Scale-Up Programme
Read more ›
European Parliament votes for EMA in the Netherlands
Read more ›

A mission to China with Prime Minister Rutte
Read more ›

Joins us for the BIO International Convention 2018 and the Visitors Programme
BIO International Convention is back in Boston, June 4-7 2018, to celebrate history-making innovation. This is the only event where you can access the global biotech and pharma community via Partnering, attend 150 education sessions and network with 16,000+ attendees from 74 countries. Discover the next generation of cutting-edge products, therapies, and cures at events all week long at BIO 2018.
Plan your stay for BIO
Don’t wait too long with planning your trip, because hotels are filling up fast. You can meet HollandBIO and Health~Holland from June 5 - 7 at the Health~Holland Pavilion, Booth #2635. On Tuesday June 5 you will find the exhibition floor's best-visited happy hour during the drinks at the Health-Holland Pavilion. True to tradition, we will raffle the orange bike! But, there are plenty of other exciting possibilities to get the best out of your Boston trip.
June 1 - 3 Executive Training Programmes
The programme supports the activities that takes place at the partnering, investor and venture capital conferences. Each course is taught by experienced business development professionals from leading pharmaceutical and biotechnology firms, and experts from the fields of law and public relations. Click here for more information.
Massachusetts Biotech Ecosystem Tours
Explore one of the world’s leading biotechnology hubs in a full day of site visit tours throughout the local Massachusetts community. As the nation’s leader in the number of biotech R&D employees, Massachusetts is home to thousands of life sciences companies driving innovation. Attend one of the 10 site, lab, and incubator tours arranged by BIO and the Boston Life Sciences Community. Learn more about the full BIO programme via this link.
Monday June 4: Welcome reception
Forget your jetlag, join the Dutch for drinks in the afternoon and get this BIO started! Location TBA
Dutch Visitors Programme BIO Boston
Would you like to experience to visit relevant organisations in and around Boston? Join the Dutch Visitors Programme (June 3 – June 7) led by chairman a.i. of the Top Sector LSH Hans Schikan and gain great insights. Meet relevant partners at the BIO and the companies we visit, like research institutes, incubators and accelerators, innovation hubs and pharmaceutical companies. Start-ups joining this mission can receive a substantial discount of € 1.500,- through the SIB Voucher. To find out if you are eligible for this discount, click here (in Dutch). The Dutch Visitors Programme is open for all Dutch Life Sciences & Health organisations in the Biotechnology & Pharmaceuticals sector. For more information about the programme, click here.
The Netherlands remains the best country according to the European Health Consumer Index
The Dutch healthcare system scores the highest in Europe in the Euro Health Consumer Index (EHCI). This is the annual comparison of health systems. In the category of medicines, the Netherlands scores 8.9.
The EHCI assesses European countries for six different categories. One is about medicines. The category of medicines consists of six parts. In this category, the Netherlands scores 'good' and 'neutral' in four parts. Furthermore, in this category the Netherlands scores 89 out of 100 points. With this, the Netherlands shares first place with Germany.
Antibiotics
In the category medicines, the EHCI looks at the use of antibiotics. In the Netherlands, antibiotic use is the lowest in Europe. Increasing antibiotic resistance, bacteria insensitive to antibiotics, means it is particularly important that doctors are cautious in prescribing antibiotics. The EHCI study shows that people in the Netherlands are well aware that antibiotics do not help against colds and flu.
Voice of the patient
The Swedish Health Consumer Powerhouse publishes a new edition of the EHCI every year. According to the EHCI, the Dutch healthcare system has few weak spots. The voice of the patient is heard and access to the care is well arranged. The researchers note that 'decisions in healthcare are taken to an unusually high degree by medical professionals with the cooperation of patients and not by the government'.
Best health care system
The Netherlands has been ranked first in the EHCI since 2008. As the Netherlands is also structurally in the top 4 of specialist indexes on diabetes, HIV and hepatitis, the EHCI no longer speaks exclusively of the most consumer-friendly care system. They indicate that it can now be said that in general the Netherlands has the best healthcare system in Europe.

European Parliament votes for EMA in the Netherlands
The European Parliament has approved the relocation of the European Medicine Agency (EMA) to Amsterdam, provided that the authorities submit quarterly reports on the progress of the construction of the new office in Amsterdam’s Zuidas business district. A large majority of the parliament (507 against 112) voted for the Netherlands as EMA's new location.
Employees
The employees working at the agency also prefer Amsterdam above the other candidate cities. According to a survey taken by the EMA itself, 81% of the employees are willing to move to the Netherlands when the agency is relocated to Amsterdam. As the EMA counts almost 900 employees this is a major asset for the Netherlands.
According to Jan Huitema (VVD): “The Netherlands has shown its ability to accommodate EMA. It is not for nothing that Amsterdam is the employees’ preference.’’
EMA building
The EMA will move from London to Amsterdam by no later than 30 March 2019, but the first employees are likely to come to the Dutch capital earlier. The employees will be temporarily accommodated in the Spark building in Amsterdam until construction of the new building has been completed in November 2019.
Click here for EMA's Tracking Tool, showing the main milestones and deliverables for the Agency's move to Amsterdam.
Read all about the advantages of setting up your business in the Netherlands in the special bidbook for the biopharmaceutical industry. It demonstrates all the reasons why the Netherlands is a top location for pharmaceutical operations for those biopharmaceutical companies that are interested in locating their company in the Netherlands. Read the bidbook


Join the second edition of the Global Scale-Up Programme to expand and scale-up your business
24-25 Sept 2018, Amsterdam / 24-30 October 2018, California
The Global Scale-up Program, sponsored by main partner Health~Holland, is developed to help ventures expand and scale-up their businesses. During this programme entrepreneurs will obtain the knowledge and skills necessary for scaling-up a life sciences and medical devices venture. They start with a baseline assessment to compare their venture to US and European competitors. Based on the outcome, they will receive one-on-one mentoring to guide them with their decision-making on how to increase the value of their venture. Subsequently they will embark upon an investment journey to Southern California.
This course is developed for members of a venture team, one to three years from launching their product, moving their focus from technical innovation to business preparation. In this phase business insights are becoming increasingly important for success. This leads to challenges such as: What makes a company attractive for investors? How does one prepare best for a next-investor round? In which countries to launch a product? How to prepare for marketing the product? What should the management team look like in a business environment?
To address these challenges, the programme will allow ventures to:
· Learn how to strategize their market positioning and launch within one to three years.
· Learn how to select optimal market entry points and the optimal partner or investor.
· Experience different techniques for venture branding and communication.
· Scan US market exploration in key regions for their venture.
· Learn to think as a global formal investor and translate this to their own venture proposition.
· Improve their pitching skills and subsequently pitch to US investors to obtain funding.
Register before 1 June to participate in the intake day (15 June). To register or if you have any questions, please email r.jongkind@hyphenprojects.nl. For more information please visit the Global Scale-Up Program website.
The Global Scale-up Program is powered by main partner Health~Holland (Top Sector Life Sciences & Health).

A mission to China: the largest trade mission of Prime Minister Rutte ever
When they came together in a huge ballroom of the Ritz-Carlton in the South China city of Guangzhou, the Dutch trade delegation was so big this time that they hardly fitted together on a group photo. "It is the biggest trade mission that I have ever led as prime minister," says Mark Rutte. "At the trade dinner tonight, we will welcome six hundred guests. It is really huge."
From the 9 to 14 April 2018 the Dutch Embassy in Beijing, theDutchConsulate in Guangzhou, the Dutch Consulate in Chongqing, NBSO Chengdu, the Netherlands Enterprise Agency (RVO) and the Task Force Health Care organised a Life Sciences & Health (LSH) programme as part of the economic trade mission to China.
In total, 29 organisations and institutions, ranging from innovative product developers to universities and even clinics and inbound medical travel organisations participated in the LSH programme. Led by Minister for Medical Care and Sport,Bruno Bruins, the Dutch delegation enjoyed a five-day programme throughout China. A large number of field visits and seminars in Beijing, Guangzhou and Chengdu provided excellent opportunities to meet new relevant business contacts, learn more about the Chinese LSH sector and foster Chinese-Dutch partnerships.
The presence of Dutch and Chinese high-level representatives also provided an excellent opportunity for some participating companies and organisations to sign agreements with their Chinese counterparts. TFHC Partner Ziuz Medical for example,has signed a distribution agreement for China with DIH Technologies Co. DIH is a strong partner for them with an impressive track record in pharmacy automation in China.
Together ZiuZ and DIH will help to make medicine dispensing in China safer and more efficient.
Another example is Maastro Clinic, which signed a Memorandum of Understanding with a Chinese counterpart for the training of medical specialists and staff in radiotherapy. The high level of knowledge and expertise of MAASTRO medical specialists and years of experience in training radiotherapist oncologists, clinical physicists and radiotherapeutic laboratory technicians are reasons for the Chinese to choose MAASTRO as a training institute.
Many opportunities for Sino-Dutch cooperation in the field of LSH are being sown and harvested in these fruitful times, with China being a focus country for the Top Sector LSH and another health delegation (elderly care) already flying out to China at the beginning of May.
Interested in more international missions and incoming visits? Check out some others:
